Posted on 17 Tháng 7 - 2025
SOFTGEL CAPSULE PRODUCTION LINE
Softgel Capsule Production Process (Including oval and oblong 16-shaped softgel capsules) A. Production Process Flowchart B. Production Process Description Receiving Raw Materials and Packaging for the Production Workshop: Raw materials (including active ingredients and excipients) and packaging materials imported into the company’s warehouse are subject to quality control. Only materials that meet quality standards […]
Softgel Capsule Production Process
(Including oval and oblong 16-shaped softgel capsules)
A. Production Process Flowchart
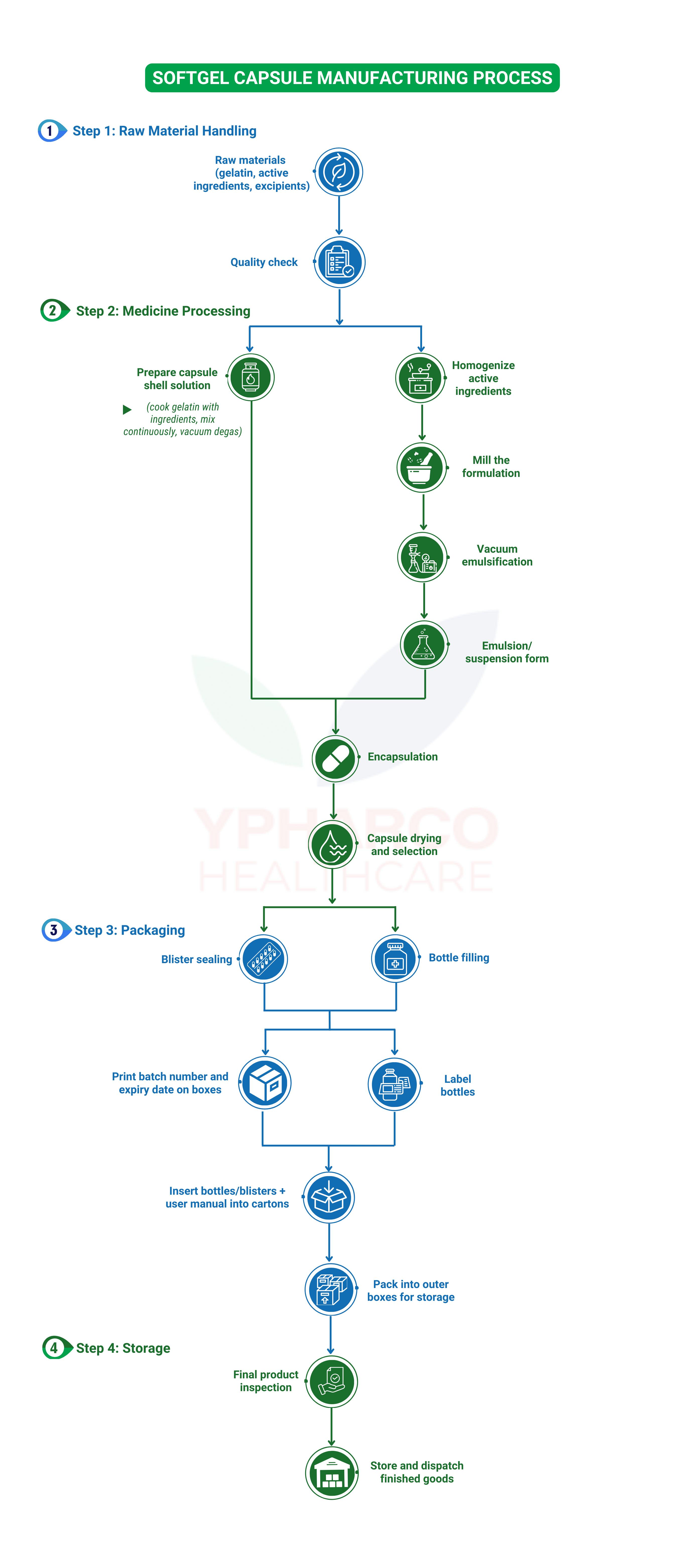
B. Production Process Description
- Receiving Raw Materials and Packaging for the Production Workshop:
Raw materials (including active ingredients and excipients) and packaging materials imported into the company’s warehouse are subject to quality control. Only materials that meet quality standards are approved for production, based on production orders and material requisition forms, under the supervision of warehouse IPC staff, production personnel, and storekeepers to ensure accurate and sufficient issuance of raw and auxiliary materials for manufacturing.
- Formulation Process:This includes gelatin cooking (capsule shell) and preparing the filling solution (active ingredients inside the capsule).
- Step 1: Gelatin Cooking
Ingredients for the capsule shell such as gelatin, sorbitol, glycerin, colorants, etc., are added into the gelatin cooking tank at a temperature of 65-75°C with purified water. The mixture is continuously stirred under vacuum pressure to create a uniform liquid without air bubbles and with moderate viscosity. The gelatin solution is then filtered through a fabric filter into a holding tank, incubated at 50-55°C for approximately 15-20 hours.
- Step 2: Intestinal fluid compounding procedure
Beeswax and palm oil are dissolved in the mixing tank using a water bath heating method. Then, the active ingredients are gradually added into the above mixture and stirred thoroughly to form a uniform liquid mass → the mixture is transferred to the grinding machine to ensure insoluble substances do not clump together and are evenly dispersed. The filling solution after grinding is transferred to the vacuum emulsifier to create a homogeneous suspension/emulsion → then placed into an incubation tank at a temperature of 28–30°C.
- Step 3: Capsule Formation
Gelatin shell and filling solution are fed into the encapsulation machine. Set the intermediate tank temperature from 45–55°C, capsule-forming drum temperature from 50–60°C, sealing knife temperature from 70–80°C, capsule shell thickness from 0.78–0.82 mm, sealing pressure from 100–150 kg/cm², and drying drum rotation reversal and capsule wiping time to 5 minutes. → Start the encapsulation machine and dry the capsules.
This step requires the capsules to have sufficient weight, properly sealed without edge openings, and capsule shell thickness must not be too thin or too thick.
- Step 4: Capsules exiting the drying drum are spread thinly on drying trays, then placed in a cold drying room and sorted at 20°C with humidity below 20% for a minimum of 48 hours before being collected and moved to the quarantine storage room to await primary packaging.
- Step 5: Primary Packaging
+ If the product requires bottling, the capsules are transferred to the counting conveyor, followed by sealing with a foil seal and capping.
+ If the product requires blister packaging, the capsules are transferred to the blister packing machine.
- Secondary Packaging Stage:
- Labeling bottles
- Inkjet printing batch number and expiration date on product cartons and master carton labels
- Placing bottles/blisters together with one instruction leaflet into each carton, closing the carton, and sealing it with a tamper-evident label
- Shrink-wrapping the carton, then placing it into the shipping box
- Moving finished products into the designated quarantine storage area
- Product Release:
Finished products are only released from the production facilities to warehouse once they have passed quality control and meet all required standards.
Softgel Capsule Production Technology
At Ypharco Healthcare, we believe that investing in technology means investing in quality and in the success of our partners. Our manufacturing facility is equipped with a highly automated, modern, and synchronized system of machinery imported from leading industrial countries.
Each piece of equipment in the production line plays a vital role in creating perfect softgel capsules that strictly comply with the most rigorous standards such as GMP, FDA, and ISO 22000:2018.
Let’s take a closer look at the advanced machinery and equipment that have built Ypharco Healthcare’s reputation.

Gelatin Melting Tank
The creation of a perfect softgel capsule begins at its core — the gelatin shell. At Ypharco Healthcare, our dedicated gelatin melting tank is made from pharmaceutical-grade 316L stainless steel, ensuring absolute purity.
A multi-layer heating system precisely controlled by sensors allows the gelatin mass to melt at the ideal temperature. Combined with a slow-speed agitator, this process produces a clear, homogeneous solution completely free of air bubbles.
This meticulous preparation is the foundation for a softgel shell that is elastic, durable, and capable of fully protecting the active ingredients — while also enhancing the product’s visual appeal for our valued partners.
Suspension Grinder
For active ingredients in suspension form, the uniformity of the fill solution is a key factor in determining product effectiveness. The high-performance suspension grinder at our facility applies intense mechanical shear force to break down and disperse active particles to micrometer size.
This process ensures absolutely even distribution of active ingredients throughout the entire production batch, preventing sedimentation. More importantly, optimizing particle size directly enhances bioavailability, allowing the body to absorb active compounds more quickly and efficiently.
Vacuum Mixing Tank
Sensitive active ingredients such as Omega-3, Vitamin E, or Coenzyme Q10 are highly vulnerable to oxidation. Understanding this, Ypharco Healthcare utilizes a mixing tank system equipped with a high-capacity vacuum pump. Before mixing, all air is evacuated from the tank to create an ideal anaerobic environment.
In this oxygen-free setting, the components of the fill formulation are blended thoroughly without any contact with air. This technology not only preserves the full potency of active ingredients and prevents product degradation, but also ensures precise dosage in every capsule by completely eliminating air bubbles.
Encapsulation Machine
This is the technological heart of the softgel production line, where precision and efficiency are top priorities. Operating on an advanced Rotary Die platform, our fully automatic encapsulation machine seamlessly integrates three core processes: forming two gelatin ribbons, precisely dosing the fill formulation using high-accuracy differential piston pumps, and sealing the capsules with a heated wedge system.
The result is millions of softgel capsules that are uniform in shape, weight, and active content — a strong testament to Ypharco Healthcare’s commitment to consistent quality at an industrial production scale.
Drying Drum System
After forming, softgel capsules undergo a preliminary drying phase to set their shape and remove surface moisture. Our drying drum system consists of multiple rotating drums designed to gently and continuously move the capsules.
A precisely controlled airflow in both temperature and volume evenly contacts all surfaces of the capsules. This process effectively prevents sticking and ensures that each capsule has a smooth, glossy surface free of defects, ready for the final drying stage in specialized drying rooms.
Capsule Counting Machine
Accuracy in product quantity is not only a regulatory requirement but also a sign of respect for consumers. At Ypharco Healthcare, our automatic capsule counting system utilizes high-sensitivity photoelectric sensor technology, allowing for 99.9% accuracy in counting the number of capsules dispensed into each bottle.
This technology completely eliminates human error. With multiple counting channels operating simultaneously, it ensures outstanding packaging efficiency — enabling rapid fulfillment of large-volume orders while maintaining absolute transparency and precision.
Heat Sealing Machine
The protective seal on each bottle is Ypharco Healthcare’s final assurance of product safety and integrity. We apply high-frequency electromagnetic induction technology for seal film application. This process does not require direct contact. Electromagnetic energy heats the aluminum foil layer inside the bottle cap, causing it to melt and seal tightly to the bottle mouth, creating an airtight seal. This serves both as a reliable tamper-evident indicator and as a critical barrier against moisture and air intrusion, helping to preserve product quality throughout distribution and use.
Blister Packaging Machine
To ensure optimal protection and high convenience, Ypharco Healthcare employs an automatic blister packaging system for products that require individual packaging. The entire process is fully automated — from PVC tray forming and precise capsule placement, to aluminum foil sealing and batch number/expiration date stamping. Each capsule is sealed within separated compartment, creating a secure barrier against environmental factors. This packaging method not only preserves product quality but also reflects the brand’s professionalism and premium positioning, while providing maximum convenience for the end user.
With comprehensive investment in a modern softgel capsule production line, Ypharco Healthcare is confident in delivering products with high precision, consistent quality, minimal loss and deviation rates — fully meeting even the most stringent customer requirements.

Contact consultant
We will contact you as soon as possible

